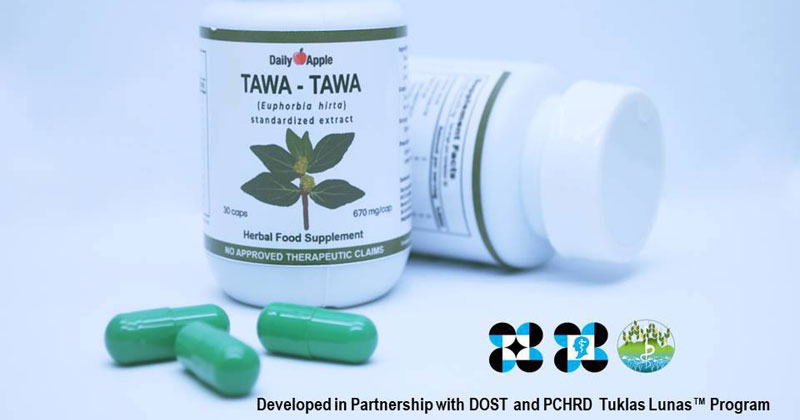
An herbal capsule made from Tawa-tawa has been approved as a supplement that helps alleviate symptoms of dengue virus.
Daily Apple Tawa-tawa herbal capsules is produced by Herbanext Laboraties Inc. based in Bago City, Negros Occidental. It is now registered and approved by the Philippine Food and Drug Administration (FDA) as a food supplement.
The capsules, with FDA Registration Number FR-4000003953662, was made from “spray-dried extract” of Tawa-tawa (Euphorbia hirta), a small herb that is widely recognized in Philippine traditional medicine as a treatment for dengue and its related symptoms.
Philip Cruz, founder and president of Herbanext, said that the use of Tawa-tawa is not a new discovery against dengue.
“It has been in traditional use for more than a century, and the most popular herbal medicine being taken throughout the country, especially in the provinces,” he said in an interview over ANC.

Herbanext said that based on their studies tawa-tawa and its active components help in alleviating dengue by increasing blood platelet count and preventing the multiplication of dengue virus in the body.
The efficacy of tawa-tawa and its active constituents against dengue is well supported and has been documented in a substantial number of studies ranging from in silico (computer-generated) simulations, in vitro (against infected cell cultures), and in vivo (in mice and people) models. It is hypothesized that there are two possible mechanisms by which tawa-tawa extracts work against dengue virus: as an anti-thrombocytopenic (prevents loss of platelets or increases platelet count), and an antiviral agent. An in silico study on quercetin, one of tawa-tawa’s active compounds was able to show that the compound inhibited the activity of several viral proteins involved in replication, even at minute concentrations. This was further supported by an in vitro study on viral cultures of dengue, wherein the compound was able to stop viral multiplication. One of the main clinical studies done on tawa-tawa, performed in India, was able to show that 70% of patients (out of a population of 125 patients) recovered from the symptoms of dengue (increased platelet count, relief from fever) after administration with tawa-tawa infusion (in addition to oral rehydration), as opposed to the group that was simply given the basic treatment protocol for dengue symptoms (i.e. oral rehydration). In terms of its anti-thrombocytopenic properties, tawa-tawa has been shown to increase platelet count in rat experimental models.
DOST Assistance
Daily Apple Tawa-tawa capsule was developed through the help of Department of Science and Technology – Philippines Council for Health Research and Development (DOST-PCHRD) Tuklas Lunas drug discovery program.

In an interview with Philippine News Agency, Secretary Fortunato dela Peña said that DOST spent P8.3 million for the equipment used by Herbanext in developing the capsule and other herbal products.
He said that Herbanext conducts studies on herbal plants that may cure headache, fever, problem in urination, among others.
“They prioritize plants or fruits that are known to be associated with illnesses based on folkloric literature,” he added.
“It is not considered medicine until it’s not tested on humans. A supplement, if you talk about safety, is something that may not cure but would not cause harm. It is safe (to use),” Dela Peña explained.
Supplements, not medicine
Department of Health (DOH) was quick to point out that the FDA-approved Tawa-tawa capsule is just a supplement and should not be used as medicine to cure or prevent dengue virus.
“At this time, it is only registered as a supplement… It means it is safe. But it has no therapeutic claims,” Health Undersecretary Rolando Enrique Domingo said. “It still has a lot of clinical trials to go through before its therapeutic value can be approved,” he added.

Domingo emphasized that those exhibiting symptoms of the mosquito-borne illness should seek immediate medical consultation.
According to Herbanext, dengue patients or those who suspect the onset of dengue should take one to two capsules three times daily until symptoms of fever improve and blood platelet count increases and stabilizes.
For those who have difficulty swallowing capsules, the contents of the capsule may be mixed in a drink with powdered milk, sugar, or honey, and fed directly.
The company targets to launch Daily Apple Tawa-tawa capsule on August 21 in their plant in Bago City. The supplement will be initially available at the Daily Apple stores in Quezon City, Bacolod, Cebu, Davao, and General Santos.
“We are contacting pharmacies to also make this product available to more people by September,” said Cruz.
DOH have already declared national dengue epidemic due to rising cases of the disease. As of August 9, there are already 160,000 cases of dengue this year, with 661 fatalities so far.